3-Mercaptopyruvate sulfurtransferase supports endothelial cell angiogenesis and bioenergetics
- Abdollahi Govar, Armita Department of Anesthesiology, University of Texas Medical Branch Galveston Texas USA
- Törö, Gábor Department of Anesthesiology, University of Texas Medical Branch Galveston Texas USA
- Szaniszlo, Peter Department of Otolaryngology, University of Texas Medical Branch Galveston Texas USA
- Pavlidou, Athanasia Laboratory of Pharmacology, Faculty of Pharmacy, National and Kapodistrian University of Athens Athens Greece
- Bibli, Sofia-Iris Institute for Vascular Signalling, Centre for Molecular Medicine, Goethe University Frankfurt Germany
- Thanki, Ketan Department of Surgery, University of Texas Medical Branch Galveston Texas USA
- Resto, Vicente A. Department of Otolaryngology, University of Texas Medical Branch Galveston Texas USA
- Chao, Celia Department of Surgery, University of Texas Medical Branch Galveston Texas USA
- Hellmich, Mark R. Department of Surgery, University of Texas Medical Branch Galveston Texas USA
- Szabo, Csaba Department of Anesthesiology, University of Texas Medical Branch Galveston Texas USA - Chair of Pharmacology, Department of Science and Medicine, University of Fribourg, Switzerland
- Papapetropoulos, Andreas Laboratory of Pharmacology, Faculty of Pharmacy, National and Kapodistrian University of Athens Athens Greece - Clinical, Experimental Surgery and Translational Research CenterBiomedical Research Foundation of the Academy of Athens Athens Greece
- Módis, Katalin Department of Anesthesiology, University of Texas Medical Branch Galveston Texas USA - Department of Surgery, University of Texas Medical Branch Galveston Texas USA
-
14.01.2020
Published in:
- British Journal of Pharmacology. - 2020, vol. 177, no. 4, p. 866–883
English
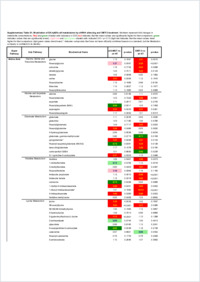
During angiogenesis, quiescent endothelial cells (ECs) are activated by various stimuli to form new blood vessels from pre‐existing ones in physiological and pathological conditions. Many research groups have shown that hydrogen sulfide (H2S), the newest member of the gasotransmitter family, acts as a proangiogenic factor. To date, very little is known about the regulatory role of 3‐mercaptopyruvate sulfurtransferase (3‐MST), an important H2S‐producing enzyme in ECs. The aim of our study was to explore the potential role of 3‐MST in human EC bioenergetics, metabolism, and angiogenesis.Experimental Approach: To assess in vitro angiogenic responses, we used EA.hy926 human vascular ECs subjected to shRNA‐mediated 3‐MST attenuation and pharmacological inhibition of proliferation, migration, and tube‐like network formation. To evaluate bioenergetic parameters, cell respiration, glycolysis, glucose uptake, and mitochondrial/glycolytic ATP production were measured. Finally, global metabolomic profiling was performed to determine the level of 669 metabolic compounds.Key Results: 3‐MST‐attenuated ECs subjected to shRNA or pharmacological inhibition of 3‐MST significantly reduced EC proliferation, migration, and tube‐like network formation. 3‐MST silencing also suppressed VEGF‐induced EC migration. From bioenergetic and metabolic standpoints, 3‐MST attenuation decreased mitochondrial respiration and mitochondrial ATP production, increased glucose uptake, and perturbed the entire EC metabolome.Conclusion and Implications: 3‐MST regulates bioenergetics and morphological angiogenic functions in human ECs. The data presented in the current report support the view that 3‐MST pathway may be a potential candidate for therapeutic modulation of angiogenesis.
- Faculty
- Faculté des sciences et de médecine
- Department
- Médecine 3ème année
- Language
-
- English
- Classification
- Biological sciences
- License
-
License undefined
- Identifiers
-
- RERO DOC 328207
- DOI 10.1111/bph.14574
- Persistent URL
- https://folia.unifr.ch/unifr/documents/308464
Other files
Statistics
Document views: 198
File downloads:
- pdf: 447
- Supplementary material: 200