Characterization of BRPMBL the bleomycin resistance protein associated with the carbapenemase NDM
- Dortet, Laurent Bacteriology-Hygiene unit, Assistance Publique/Hôpitaux de Paris, Bicêtre Hospital, Le Kremlin-Bicêtre, France - EA7361 (Structure, dynamic, function and expression of broad spectrum β-lactamases), Paris-Sud University, LabEx Lermit, Faculty of Medicine, Le Kremlin-Bicêtre, France - Associated French National Reference Center for Antibiotic Resistance: Carbapenemase-producing Enterobacteriaceae, Le Kremlin-Bicêtre, France
- Girlich, Delphine EA7361 (Structure, dynamic, function and expression of broad spectrum β-lactamases), Paris-Sud University, LabEx Lermit, Faculty of Medicine, Le Kremlin-Bicêtre, France
- Virlouvet, Anne-Laure Bacteriology-Hygiene unit, Assistance Publique/Hôpitaux de Paris, Bicêtre Hospital, Le Kremlin-Bicêtre, France
- Poirel, Laurent Emerging Antibiotic Resistance Unit, Medical and Molecular Microbiology, Department of Medicine, University of Fribourg, Switzerland - INSERM European Unit (LEA Paris, IAME, France), University of Fribourg, Switzerland
- Nordmann, Patrice Emerging Antibiotic Resistance Unit, Medical and Molecular Microbiology, Department of Medicine, University of Fribourg, Switzerland - INSERM European Unit (LEA Paris, IAME, France), University of Fribourg, Switzerland - University hospital and University of Lausanne, Switzerland
- Iorga, Bogdan I. Institut de Chimie des Substances Naturelles, CNRS UPR 2301, LabEx LERMIT, Gif-sur-Yvette, France
- Naas, Thierry Bacteriology-Hygiene unit, Assistance Publique/Hôpitaux de Paris, Bicêtre Hospital, Le Kremlin-Bicêtre, France - EA7361 (Structure, dynamic, function and expression of broad spectrum β-lactamases), Paris-Sud University, LabEx Lermit, Faculty of Medicine, Le Kremlin-Bicêtre, France
-
03.01.2017
Published in:
- Antimicrobial Agents and Chemotherapy. - 2017, vol. 61, no. 3, p. e02413-16
English
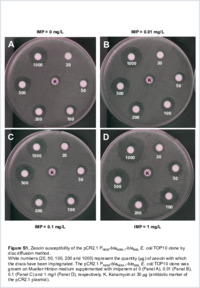
The metallo-β-lactamase NDM-1 is among the most worrisome resistance determinants and is spreading worldwide among Gram-negative bacilli. A bleomycin resistance gene, bleMBL, downstream of the blaNDM-1 gene has been associated with resistance almost systematically. Here, we characterized the corresponding protein, BRPMBL, conferring resistance to bleomycin, an antitumoral glycopeptide molecule. We have determined whether the expression of the blaNDM-1-bleMBL operon is inducible in the presence of carbapenems and/or bleomycin-like molecules using quantitative reverse transcription-PCR (qRT-PCR), determination of imipenem and zeocin MICs, and carbapenemase-specific activity assays. We showed that the blaNDM- 1-bleMBL operon is constitutively expressed. Using electrophoretic mobility shift and DNA protection assays performed with purified glutathione S- transferase (GST)-BRPMBL, we demonstrated that BRPMBL is able to bind and sequester bleomycin-like molecules, thus preventing bleomycin-dependent DNA degradation. In silico modeling confirmed that the mechanism of action required the dimerization of the BRPMBL protein in order to sequester bleomycin and prevent DNA damage. BRPMBL acts specifically on bleomycin-like molecules since cloning and expression of bleMBL in Staphyloccoccus aureus did not confer cross-resistance to any other antimicrobial glycopeptides such as vancomycin and teicoplanin.
- Faculty
- Faculté des sciences et de médecine
- Department
- Médecine 3ème année
- Language
-
- English
- Classification
- Biological sciences
- License
-
License undefined
- Identifiers
-
- RERO DOC 287914
- DOI 10.1128/AAC.02413-16
- Persistent URL
- https://folia.unifr.ch/unifr/documents/305288
Other files
Statistics
Document views: 152
File downloads:
- pdf: 274
- Supplementary material: 127